Organ Transplant Rejection Medication Market Size
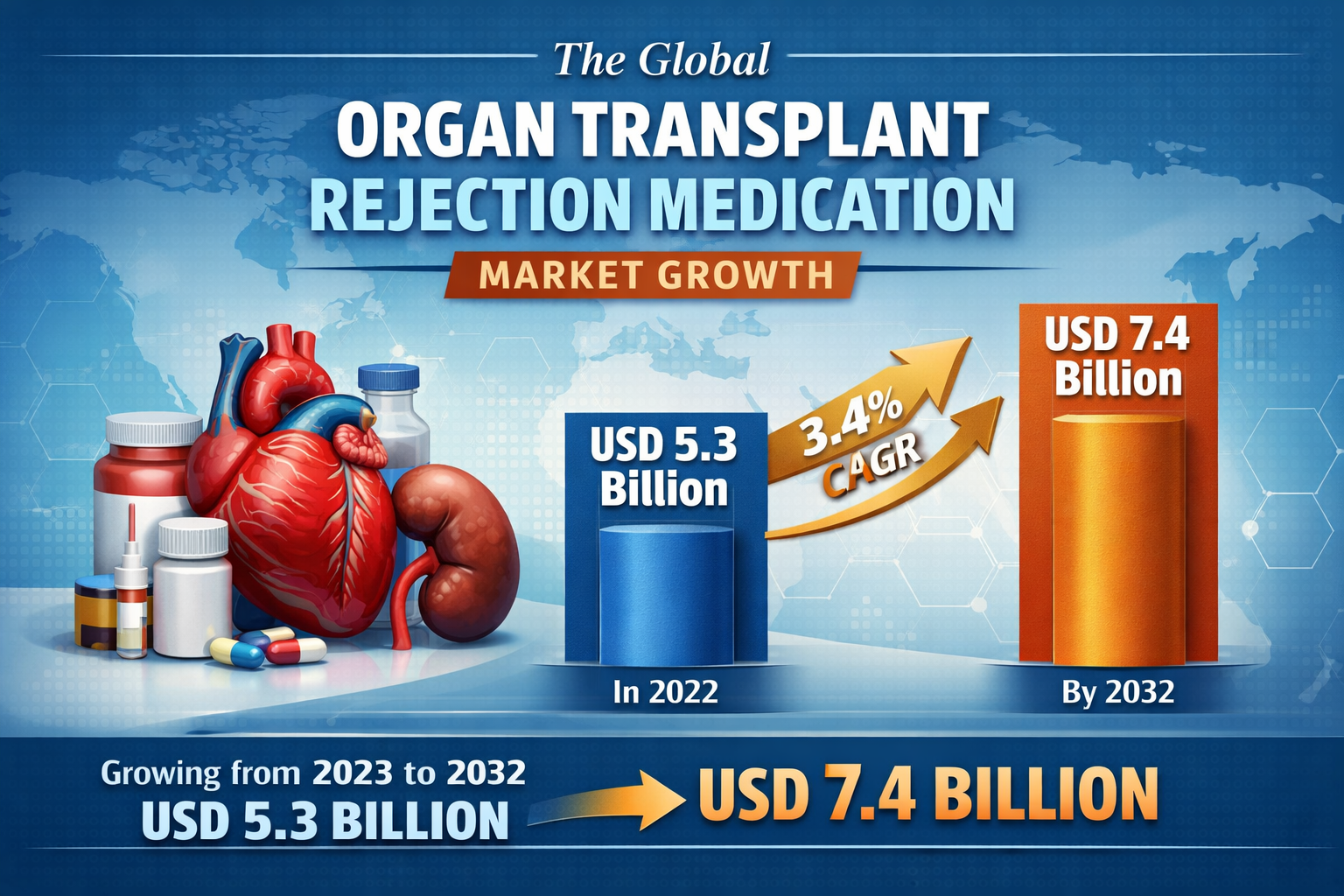
The Organ Transplant Rejection Medication Market was valued at USD 5.3 billion in 2022 and is projected to grow to USD 7.4 billion by 2032 at a CAGR of 3.4%.
What Is the Organ Transplant Rejection Medication Market?
The Organ Transplant Rejection Medication Market refers to the global industry involved in the development, production, and distribution of pharmaceuticals that prevent immune‑mediated rejection of transplanted organs. These medications suppress the activity of the recipient’s immune system to reduce the risk that the body will attack the new organ. They include multiple drug classes such as:
Calcineurin inhibitors
Antiproliferative agents
Corticosteroids
Antibodies and biologics
mTOR inhibitors
Each class functions by targeting different aspects of the immune response to balance suppression with safety and effectiveness.
Market Significance
These medications are essential for recipients of kidney, liver, heart, lung, pancreas, and other organ transplants.
Their use is typically lifelong, making them a critical and sustained component of post‑transplant care.
Growth in the market parallels increases in transplant procedures globally and advancements in immunotherapy.
Get a Free Sample: https://www.acumenresearchandconsulting.com/request-sample/3393
Market Trends
The organ transplant rejection medication market is witnessing several notable trends:
1. Personalized Medicine and Precision Approaches
Healthcare practitioners increasingly tailor immunosuppressive regimens based on genetic and molecular profiling, optimizing therapy to patient-specific immune response patterns. Personalized approaches aim to improve graft outcomes while minimizing side effects.
2. New Drug Development and Biosimilars
The introduction of biosimilars — cost‑competitive alternatives to established biologics — is reshaping treatment options. These products help broaden access and reduce treatment cost pressures in some regions.
3. Emergence of Regenerative Medicine
Research into regenerative medicine, xenotransplantation, and organ bioengineering hints at alternative strategies that could reduce dependence on traditional immunosuppressants over time. Though still largely experimental, these areas represent future growth opportunities.
Market Dynamics
Market growth in this sector is influenced by a combination of drivers, restraints, opportunities, and challenges:
Key Drivers
Rising Prevalence of Organ Failure: Chronic diseases like kidney failure, liver cirrhosis, and heart disease increase the demand for organ transplants and associated medication.
Advances in Transplant Technology: Better surgical and clinical care expands transplant eligibility and success, enlarging the medication market.
Awareness and Early Intervention: Heightened public and clinical awareness enhances early transplant care and post‑transplant management.
Restraints
High Treatment Costs: The high price of some immunosuppressants poses affordability challenges, especially in low‑income regions.
Side Effect and Complication Risks: Long‑term immune suppression increases vulnerability to infections and other health risks, impacting patient compliance and quality of life.
Opportunities
Emerging Markets: Rapid healthcare improvement in Asia‑Pacific, Latin America, and parts of Africa presents new patient populations and market expansion potential.
Innovative Therapies: Continued R&D into targeted immunosuppressants and combination therapies offers potential for safer, more effective drug options.
Challenges
Regulatory Barriers: Strict regulatory requirements can slow approval and market entry for novel therapies.
Patent Expirations: While generics increase access, they may also reduce manufacturer incentives for innovation if not well compensated.
Regional Analysis
Geographically, the market shows varied performance and potential:
North America
Accounts for the largest share of the global market (>43%).
Advanced healthcare infrastructure, high transplant procedure volumes, and strong R&D activities underpin this leadership position.
Asia‑Pacific
Expected to exhibit the fastest CAGR through 2032 due to improving healthcare access, rising chronic disease burden, and increasing transplant awareness.
Europe, Latin America, Middle East & Africa
Each region benefits from different factors such as public health initiatives, regulatory frameworks, and investment in medical facilities, with Europe remaining a significant market due to established healthcare systems.
Recent Developments
FDA Approval of Novel Transplant‑Associated Therapy: In late 2025, the U.S. FDA approved a monoclonal antibody drug to treat transplant‑associated complications, illustrating ongoing innovation in the transplant drug landscape.
Clinical and Regulatory Approvals: Expanded indications for existing drugs and generic approvals continue to shape treatment options and competitive dynamics.
Conclusion
The Organ Transplant Rejection Medication Market remains a vital and steadily growing segment of the global pharmaceutical landscape. Fueled by advances in transplant surgeries, personalized medicine, expanding patient populations, and emerging markets, its evolution is propelled by both clinical need and innovation. While challenges such as cost and safety persist, opportunities in new therapies and broader global access signal a robust future for this market.
To Get Detailed Overview, Contact Us: https://www.acumenresearchandconsulting.com/contact-us