Table of Contents
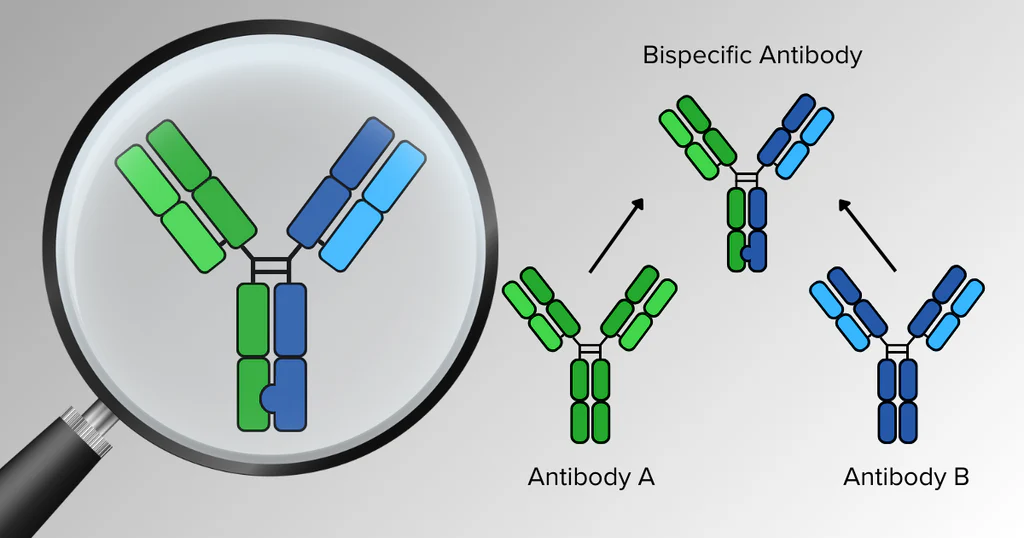
ToggleBispecific Antibodies Market Size
The global bispecific antibody market was valued at approximately USD 554 million in 2022 and is projected to reach nearly USD 1,084 million by 2032, expanding at a CAGR of around 7.1%, reflecting strong clinical adoption and pipeline expansion across multiple therapeutic indications.
Growth Factors Driving the Bispecific Antibodies Market
The growth of the bispecific antibodies market is primarily supported by the rapid expansion of immuno-oncology research, increasing regulatory approvals for next-generation antibody therapeutics, rising global cancer incidence, and strong investment in biologics manufacturing platforms; additionally, pharmaceutical companies are accelerating clinical development programs with more than 200 bispecific antibody candidates currently in clinical or preclinical development pipelines worldwide, while favorable regulatory policies, growing adoption in autoimmune and infectious disease treatment, and advances in antibody engineering technologies such as T-cell engagement platforms and dual checkpoint inhibitors are further strengthening commercialization prospects, alongside increased collaboration between biotechnology firms and academic research institutions that are accelerating translational innovation and expanding therapeutic indications globally.
Leading Trends and Their Impact on Market Evolution
Expansion of T-Cell Engager Platforms
T-cell engager bispecific antibodies represent the fastest-growing therapeutic class due to their ability to redirect immune cells toward malignant targets. These therapies are redefining treatment approaches for hematologic cancers and solid tumors.
Impact:
They are enabling personalized oncology therapies with higher response rates and reduced toxicity compared with chemotherapy combinations.
Get a Free Sample: https://www.acumenresearchandconsulting.com/request-sample/3334
Integration with Combination Immunotherapy Pipelines
Bispecific antibodies are increasingly integrated with immune checkpoint inhibitors and CAR-T therapies to enhance clinical efficacy.
Impact:
This trend is expanding treatment paradigms in advanced oncology and improving patient survival outcomes.
Shift Toward Subcutaneous Delivery Formats
New subcutaneous delivery technologies are improving treatment accessibility and patient compliance compared with intravenous infusion therapies.
Impact:
This shift is accelerating adoption across outpatient oncology care settings.
Growing Focus on Autoimmune Disease Applications
While oncology remains dominant, autoimmune disease indications are emerging as important growth segments.
Impact:
Diversification beyond cancer therapeutics is strengthening long-term revenue sustainability for biologics developers.
Strategic Collaborations and Licensing Agreements
Partnerships between biotechnology innovators and pharmaceutical companies are expanding clinical pipelines rapidly.
Impact:
Collaborative innovation is accelerating regulatory approvals and reducing development timelines.
Successful Global Examples of Bispecific Antibody Commercialization
Several bispecific antibody therapies have demonstrated strong clinical and commercial success globally:
- Blinatumomab (Amgen) revolutionized treatment for acute lymphoblastic leukemia
- Mosunetuzumab (Roche) demonstrated strong efficacy in lymphoma therapy
- Faricimab (Roche) transformed ophthalmology treatment pathways
- Epcoritamab (Genmab/AbbVie collaboration) advanced lymphoma immunotherapy
- Teclistamab (Johnson & Johnson) expanded multiple myeloma treatment options
These therapies illustrate the expanding therapeutic breadth of bispecific antibodies beyond hematologic malignancies into ophthalmology and autoimmune indications.
Global Regional Analysis with Government Initiatives and Policy Landscape
North America
North America dominates the global bispecific antibodies market, supported by strong biotechnology infrastructure, early regulatory approvals, and extensive clinical research investments. The United States leads global pipeline development through accelerated approval pathways and immunotherapy funding initiatives.
Government support includes:
- Breakthrough therapy designation programs
- National Cancer Institute immunotherapy funding
- National Institutes of Health biologics research initiatives
These programs are accelerating translational immunotherapy innovation and commercialization.
Europe
Europe represents the second-largest market due to strong biologics manufacturing capabilities and regulatory support from regional medicines agencies.
Key initiatives include:
- Horizon Europe biomedical innovation funding
- Pharmaceutical strategy modernization policies
- Cross-border clinical research collaboration frameworks
Countries such as Germany, Switzerland, and the United Kingdom are leading antibody engineering research.
Asia-Pacific
Asia-Pacific is the fastest-growing regional market driven by expanding oncology incidence rates, government healthcare modernization programs, and biologics manufacturing investments.
Major policy drivers include:
- China’s biologics innovation funding programs
- Japan’s regenerative medicine regulatory acceleration pathways
- India’s biotechnology sector expansion incentives
These initiatives are strengthening domestic antibody manufacturing capabilities.
Latin America
Latin America is experiencing gradual adoption supported by expanding oncology treatment infrastructure and improving biologics accessibility programs. Brazil and Mexico are emerging as regional biotechnology hubs with growing biologics clinical trial participation.
Middle East & Africa
Middle East and Africa markets remain at an early stage but are expanding due to rising oncology investments and increasing biologics imports through public healthcare systems, particularly in Gulf Cooperation Council countries and South Africa.
Company Profiles: Key Market Participants
Regeneron Pharmaceuticals, Inc.
Specialization:
Monoclonal and bispecific antibody therapeutics for oncology, ophthalmology, and inflammatory diseases
Key Focus Areas:
- Immuno-oncology
- Retinal disease treatment
- Immune-mediated disorders
Notable Features:
- Proprietary VelociSuite antibody engineering platform
- Strong biologics discovery capabilities
- Expanding oncology pipeline leveraging dual-target platforms
2024 Revenue:
Approximately USD 13.1 billion
Market Share:
Moderate but rapidly expanding presence in next-generation bispecific antibody platforms
Global Presence:
North America, Europe, Asia-Pacific
Amgen Inc.
Specialization:
Biologics-based oncology and rare disease therapeutics
Key Focus Areas:
- Hematologic malignancies
- T-cell engager therapies
- Precision biologics development
Notable Features:
- Pioneer of BiTE antibody technology
- Strong pipeline of immune-cell engager therapies
- Extensive oncology commercialization network
2024 Revenue:
Approximately USD 33.4 billion
Market Share:
Strong leadership position in T-cell engager bispecific antibody segment
Global Presence:
North America, Europe, Asia-Pacific, Latin America
Roche Holding AG
Specialization:
Oncology biologics, diagnostics integration, and ophthalmology therapeutics
Key Focus Areas:
- Cancer immunotherapy
- Dual-target antibody development
- Precision diagnostics integration
Notable Features:
- Strong ophthalmology bispecific antibody portfolio
- Leadership in lymphoma-focused bispecific therapies
- Integrated diagnostics-therapeutics ecosystem
2024 Revenue:
Approximately USD 68 billion (group revenue)
Market Share:
Major global leader in oncology bispecific therapeutics
Global Presence:
Operations across more than 100 countries worldwide
Genentech, Inc.
Specialization:
Biotechnology innovation subsidiary of Roche focusing on oncology and immunology
Key Focus Areas:
- Oncology antibody engineering
- Immune checkpoint targeting
- Personalized medicine platforms
Notable Features:
- Leading antibody discovery innovation center
- Strong translational research pipeline
- Integrated global clinical trial ecosystem through Roche
2024 Revenue:
Included within Roche pharmaceutical division revenue structure
Market Share:
Significant contributor to Roche’s oncology biologics leadership
Global Presence:
North America research leadership with global commercialization support
Novartis AG
Specialization:
Innovative biologics and precision therapeutics
Key Focus Areas:
- Oncology therapeutics
- Autoimmune disease therapies
- Advanced biologics platforms
Notable Features:
- Expanding antibody engineering capabilities
- Strategic pipeline transformation toward next-generation biologics
- Strong investment in precision medicine platforms
2024 Revenue:
Approximately USD 51.7 billion
Market Share:
Growing presence in targeted biologics and immunotherapy segments
Global Presence:
Operations across more than 140 countries
Key Market Trends
- Expansion of immune-cell engager therapeutics
- Increasing regulatory approvals globally
- Rising investment in antibody engineering technologies
- Pipeline diversification beyond oncology
- Integration with next-generation precision medicine platforms
Market Drivers
- Rising cancer prevalence globally
- Growth in biologics research and development investment
- Increasing regulatory support for innovative therapeutics
- Expanding clinical pipeline activity
- Strong adoption in hematologic malignancies
Market Impact Analysis
Bispecific antibodies are transforming therapeutic delivery by enabling:
- Dual-target precision therapy
- Reduced treatment resistance
- Improved clinical response rates
- Simplified treatment regimens
- Enhanced immune system engagement
Their integration into oncology pipelines is reshaping biologics market competitiveness globally.
Market Challenges
Despite strong growth potential, the market faces several barriers:
- High manufacturing complexity
- Regulatory approval challenges
- Expensive clinical trial requirements
- Limited large-scale production infrastructure
- Reimbursement constraints in emerging markets
Market Opportunities
Significant opportunities exist in:
- Autoimmune disease therapeutics
- Ophthalmology treatment expansion
- Combination immunotherapy platforms
- Emerging markets biologics adoption
- Next-generation antibody engineering technologies
- AI-assisted biologics discovery platforms
Future Outlook
The future outlook for the bispecific antibodies market remains exceptionally strong as pharmaceutical companies continue expanding antibody engineering platforms and clinical pipelines targeting oncology, autoimmune disorders, infectious diseases, and ophthalmology applications. The integration of artificial intelligence in biologics discovery, improvements in antibody stability and manufacturability, and increasing regulatory approvals across major healthcare markets are expected to accelerate commercialization timelines significantly over the next decade. With continued investment from biotechnology innovators and global pharmaceutical leaders, bispecific antibodies are expected to transition from niche oncology therapeutics into mainstream precision medicine platforms, positioning them as one of the most strategically important biologics segments shaping the future of targeted therapy worldwide.
To Get Detailed Overview, Contact Us: https://www.acumenresearchandconsulting.com/contact-us